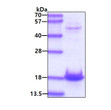
Human alpha-Synuclein (A30P) protein (Recombinant) (No-Tag) (STJP019767)
SPECIFICATIONS
HostE.coli
ImmunogenHuman
General Information
| Short Description | Recombinant-Human alpha-Synuclein (A30P)-No-Tag protein was developed from e.coli and has a target region of No-Tag. For use in research applications. |
| Applications | SDS-PAGE |
| Host | E.coli |
| Note | STRICTLY FOR FURTHER SCIENTIFIC RESEARCH USE ONLY (RUO). MUST NOT TO BE USED IN DIAGNOSTIC OR THERAPEUTIC APPLICATIONS. |
Product Properties
| Concentration | 1 mg/mL |
| Formulation | Liquid in 20mM Tris-HCl buffer (pH 7.5) containing 0.1M NaCl, 10% Glycerol |
| Storage Instruction | For short term storage, keep at +2C to +8C for up to 1 week. For long term storage, aliquot and store at-20C, and avoid repeat freeze-thaw cycles. |
Target Information
| Gene Symbol | SNCA |
| Gene ID | 6622 |
| Uniprot ID | SYUA_HUMAN |
| Accession Number | NP_000336.1 |
| Immunogen | Human |
| Immunogen Region | 1-140aa |
| Immunogen Sequence | MDVFMKGLSK AKEGVVAAAE KTKQGVAEAP GKTKEGVLYV GSKTKEGVVH GVATVAEKTK EQVTNVGGAV VTGVTAVAQK TVEGAGSIAA ATGFVKKDQL GKNEEGAPQE GILEDMPVDP DNEAYEMPSE EGYQDYEPEA |
Additional Info
| Tissue Specificity | Highly expressed in presynaptic terminals in the central nervous system. Expressed principally in brain. |
| Post Translational Modifications | Phosphorylated, predominantly on serine residues. Phosphorylation by CK1 appears to occur on residues distinct from the residue phosphorylated by other kinases. Phosphorylation of Ser-129 is selective and extensive in synucleinopathy lesions. In vitro, phosphorylation at Ser-129 promoted insoluble fibril formation. Phosphorylated on Tyr-125 by a PTK2B-dependent pathway upon osmotic stress. Hallmark lesions of neurodegenerative synucleinopathies contain alpha-synuclein that is modified by nitration of tyrosine residues and possibly by dityrosine cross-linking to generated stable oligomers. Ubiquitinated. The predominant conjugate is the diubiquitinated form. Acetylation at Met-1 seems to be important for proper folding and native oligomeric structure. |
| Function | Neuronal protein that plays several roles in synaptic activity such as regulation of synaptic vesicle trafficking and subsequent neurotransmitter release. Participates as a monomer in synaptic vesicle exocytosis by enhancing vesicle priming, fusion and dilation of exocytotic fusion pores. Mechanistically, acts by increasing local Ca(2+) release from microdomains which is essential for the enhancement of ATP-induced exocytosis. Also acts as a molecular chaperone in its multimeric membrane-bound state, assisting in the folding of synaptic fusion components called SNAREs (Soluble NSF Attachment Protein REceptors) at presynaptic plasma membrane in conjunction with cysteine string protein-alpha/DNAJC5. This chaperone activity is important to sustain normal SNARE-complex assembly during aging. Also plays a role in the regulation of the dopamine neurotransmission by associating with the dopamine transporter (DAT1) and thereby modulating its activity. |
| Protein Name | Alpha-SynucleinNon-A Beta Component Of Ad AmyloidNon-A4 Component Of Amyloid PrecursorNacp |
| Database Links | Reactome: R-HSA-977225Reactome: R-HSA-9833482 |
| Cellular Localisation | CytoplasmMembraneNucleusSynapseSecretedCell ProjectionAxonMembrane-Bound In Dopaminergic NeuronsExpressed And Colocalized With Septin4 In Dopaminergic Axon TerminalsEspecially At The Varicosities |
| Alternative Protein Names | Alpha-Synuclein proteinNon-A Beta Component Of Ad Amyloid proteinNon-A4 Component Of Amyloid Precursor proteinNacp proteinSNCA proteinNACP proteinPARK1 protein |
Information sourced from Uniprot.org