| Short Description : | Recombinant-E.coli dsbG-No-Tag protein was developed from e.coli and has a target region of No-Tag. For use in research applications. |
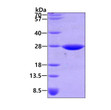
| Applications: | SDS-PAGE |
| Host: | E.coli |
| Note: | STRICTLY FOR FURTHER SCIENTIFIC RESEARCH USE ONLY (RUO). MUST NOT TO BE USED IN DIAGNOSTIC OR THERAPEUTIC APPLICATIONS. |
| Concentration: | 1 mg/mL |
| Formulation: | Liquid in 20mM Tris-HCl buffer (pH 8.0) containing 2mM EDTA, 10% Glycerol |
| Storage Instruction: | For short term storage, keep at +2C to +8C for up to 1 week. For long term storage, aliquot and store at-20C, and avoid repeat freeze-thaw cycles. |
| Accession Number: | NP_415137 |
| Immunogen: | E.coli |
| Immunogen Region: | 18-248aa |
| Immunogen Sequence: | MEELPAPVKA IEKQGITIIK TFDAPGGMKG YLGKYQDMGV TIYLTPDGKH AISGYMYNEK GENLSNTLIE KEIYAPAGRE MWQRMEQSHW LLDGKKDAPV IVYVFADPFC PYCKQFWQQA RPWVDSGKVQ LRTLLVGVIK PESPATAAAI LASKDPAKTW QQYEASGGKL KLNVPANVST EQMKVLSDNE KLMDDLGANV TPAIYYMSKE NTLQQAVGLP DQKTLNII |
| Background | Dsb proteins control the formation and rearrangement of disulfide bonds during the folding of secreted and membrane proteins in bacteria. DsbG, a member of this family, has disulfide bond isomerase and chaperone activity. So DsbG was shown to interact with refolding intermediates of chemically denatured citrate synthase and prevents their aggregation in vitro. In addition to sharing sequence homology with the thiol disulfide exchange protein DsbC, DsbG likewise was shown to form a stable periplasmic dimer and it displays an equilibrium constant with glutathione comparable with DsbA and DsbC. DsbG was found to be expressed at approximately 25% the level of DsbC. Recombinant DsbG was expressed in E. coli and purified by conventional chromatography techniques. |
Information sourced from Uniprot.org